Contractor Compliance is now a VelocityEHS company!
Find out just how easy contractor management can be. Learn More >
EHS & ESG Highlights:
MAGAZINE
Ignite Magazine
Unlock EHS & ESG Myths vs. Truths with Ignite Issue #5.
ASSESSMENT TOOL
Ergonomics Self-Assessment
Find program strengths and gaps; get customized recommendations.
Get ahead of the competition with industry-leading EHS & ESG solutions
Our seven platform solutions are specifically designed to address your key EHS- & ESG-related challenges with ease. Choose a solution below to learn how we can help you simplify compliance, drive innovation, and deliver exceptional value.
Safety
Simplify the key aspects of safety, whether you’re a safety management veteran or new to the role.
Ergonomics
Minimize the risk of MSDs and reduce the costs associated with lost productivity and injury expenses.
Control of Work
Control hazardous work activities more effectively with permit-to-work, contractor management, visitor management, and additional SaaS tools.
Chemical Management
Take command of your chemical container management, ingredient indexing, and regulatory reporting.
ESG
Make your ESG program a competitive advantage with data insights that will help you see your company in a whole new light.
Operational Risk
Gain an enterprise-level view of risk with the ability to pinpoint and evaluate all types of hazards, implement and verify risk controls, and harmonize risk management activities.
Environmental Compliance
Taking care of your people and community includes managing your air, water and waste streams. We can help.
Safety
Simplify the key aspects of safety, whether you’re a safety management veteran or new to the role.
Ergonomics
Minimize the risk of MSDs and reduce the costs associated with lost productivity and injury expenses.
Control of Work
Control hazardous work activities more effectively with permit-to-work, contractor management, visitor management, and additional SaaS tools.
Chemical Management
Take command of your chemical container management, ingredient indexing, and regulatory reporting.
ESG
Make your ESG program a competitive advantage with data insights that will help you see your company in a whole new light.
Operational Risk
Gain an enterprise-level view of risk with the ability to pinpoint and evaluate all types of hazards, implement and verify risk controls, and harmonize risk management activities.
Environmental Compliance
Taking care of your people and community includes managing your air, water and waste streams. We can help.
We make it simple for you to protect your people, profits and planet
Swiftly tackle your complex EHS & ESG challenges, create more efficient workflows, reduce costs and drive sustainable outcomes with our ActiveEHS® predictive technology solutions.
Gain Visibility
Get clear visibility into your EHS & ESG performance on a global level and drive continuous improvement across teams and time zones—all in one platform.
Leverage Our Expertise
Backed by the largest panel of certified EHS & ESG experts in the industry, we’re here to help you build the knowledge, skills and confident control that will ensure your organization’s long-term success.
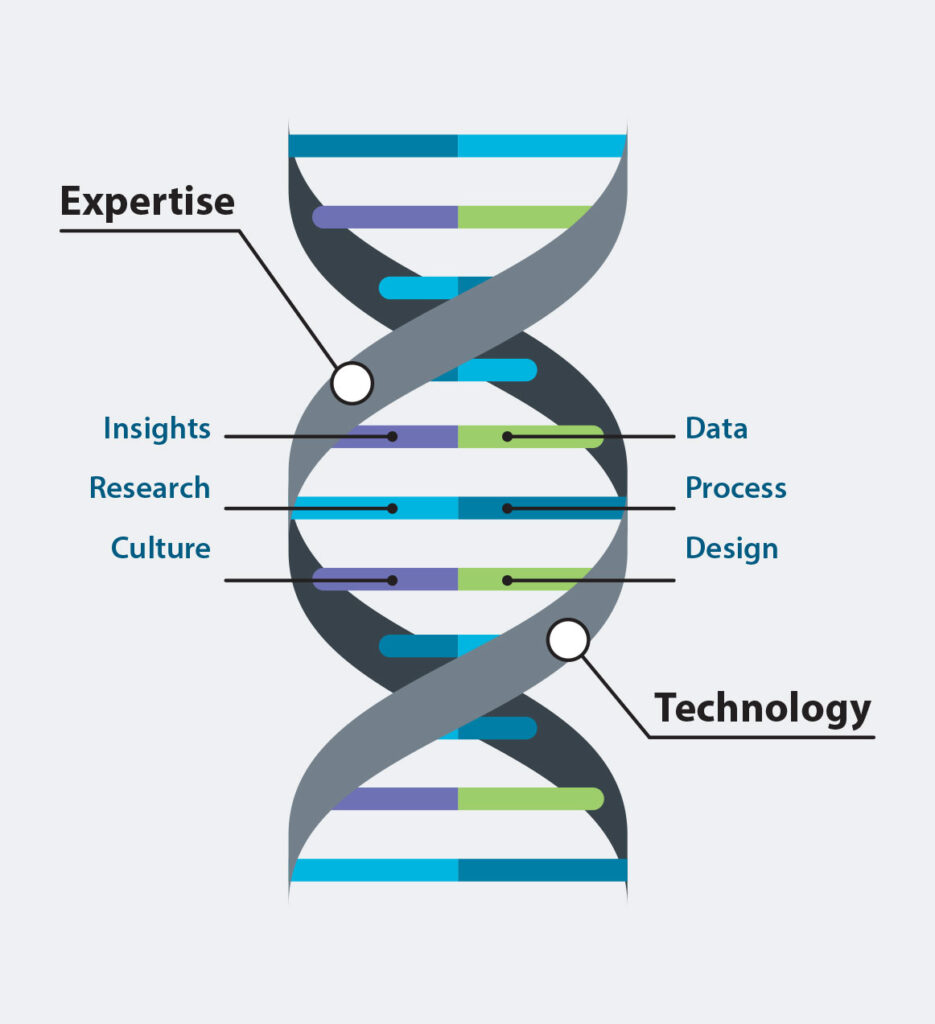
Make it Work for You
Our solutions easily adapt to your unique challenges, management strategy and workforce. No matter your desired outcomes, we’re ready to grow with you.
Predict, Prevent, Protect
Fueled by AI & machine learning, our EHS & ESG solutions help you predict obstacles, prevent costly mistakes and protect your bottom line so you can get ahead of tomorrow and stop worrying about today.
Global enterprise leaders are transforming their EHS & ESG performance with VelocityEHS
We give teams the tools they need to drive business results quickly—all while delivering EHS & ESG excellence. But don’t just take our word for it. Take a look at some of our client partners who are leading the way.

Microsoft Corporation
Toyota

Kraft Heinz

Ashland

3M
Coca-Cola
See why we’re the most trusted EHS & ESG platform in the world for Fortune 1000 companies
Users Around the World
Countries in 20+ Languages
Customer Loyalty
Learn best practices from our industry-leading experts
Check out the latest industry news, our most recent blogs, and helpful training resources that will ensure you stay ahead of the curve.

California’s ESG Disclosure Rules: What You Need to Know
ESG
California is now the first US state with ESG disclosure requirements. Here are some major takeaways about California’s ESG disclosure rules.

The Ergonomics Hit List®: A Simple Tool to Identify Issues in the Workplace
Ergonomics
In industrial environments, it’s important to take the time to properly assess the workplace for musculoskeletal disorder (MSDs) risks which can lead to sprains, st…

The Importance of Control of Work on the Path to Maturity
Control of Work
Managing contractors, temporary workers and visitors, making sure they’ve completed the necessary training, and ensuring they have the proper credentials and permit…
Ready to see VelocityEHS in action?
Request a demo today to see how we help organizations like yours gain control of their EHS & ESG strategy and empower global teams for success.